
Iryna Imago/iStock via Getty Images
On September 14, the Multidisciplinary Association for Psychedelic Studies (“MAPS”) announced positive results from a second Phase 3 trial of MDMA – also known as ecstasy – for post-traumatic stress disorder, which were published in a scientific journal.
The data into the study Naturopathy found that those given MDMA saw significant improvements in PTSD symptoms compared to those given placebo. No serious treatment-emergent adverse events or deaths have been reported.
As a result, MAPS said its subsidiary, MAPS Public Benefit Corporation, would likely file an NDA for the candidate by the end of the year. That sets the stage for possible US FDA approval in 2024.
If that were to happen, the MDMA candidate could become the first true psychedelic drug to gain FDA approval. The last agency-approved therapy with a psychedelic component came in 2019 with Johnson & Johnson’s (JNJ) Spravato (esketamine), a derivative of the dissociative hallucinogen ketamine, for treatment-resistant depression.
Psychedelic therapies for mental illness could represent a major advance in treatment for two main reasons: there is an unmet medical need for several conditions and their effects are fast-acting. For example, currently available antidepressants usually take at least a month before any treatment effect is noticeable. With Spravato, the antidepressant effect can be felt within minutes.
Psychedelic treatments are expected to become important sources of revenue for drug developers. According to a new report from Research Nester, the global market size of psychedelic drugs is expected to grow at a CAGR of more than 14% between 2023 and 2035. By the end of 2025, the market is expected to generate revenues of $12 billion, up from ~$3 billion in 2023.
The FDA has also recognized the potential benefits of psychedelics after issuing guidelines for psychedelic research in June.
 Courtesy: pharmaceutical technology
Courtesy: pharmaceutical technology
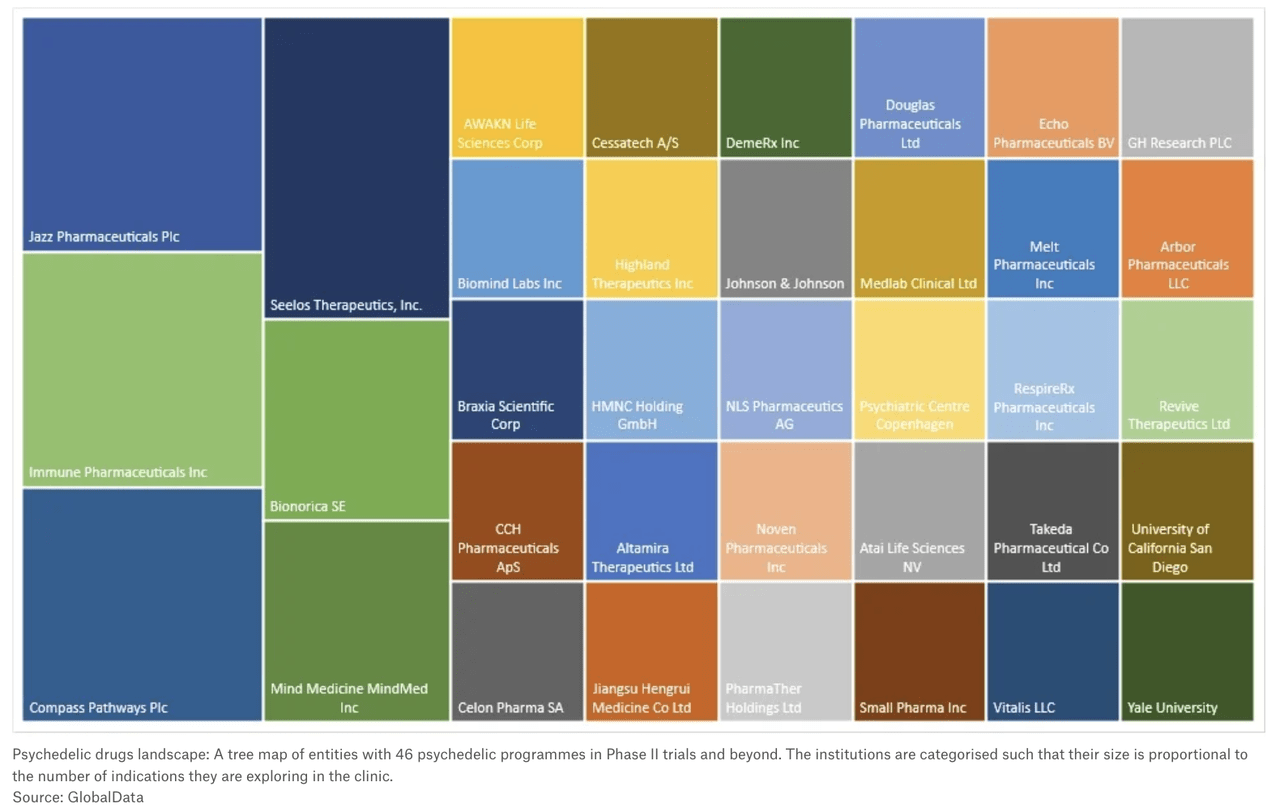
Pharmaceutical companies involved in psychedelic drug treatments are, for the most part, not household names. Publicly traded ones include COMPASS Pathways (NASDAQ:CMPS), Cybin (CYBN), Mind Medicine (NASDAQ:MNMD), Atai Life Sciences (NASDAQ:ATAI), Lucy Scientific Discovery (LSDI) Silo Pharma (SILO), Numinus Wellness (OTCQX:NUMIF), Jazz Pharmaceuticals (JAZZ), GH Research (GHRS), Bright Minds Bioscience (DRUG), 180 Life Sciences (ATNF), Seelos Therapeutics (NASDAQ: SEEL), and PharmaTher Holdings (OTCQB:PHRRF).
Even major pharmaceutical companies, like Johnson & Johnson (JNJ) and Takeda Pharmaceutucal (TAK), have dipped their toes into psychedelic development.
Although MAPS’s MDMA therapy for PTSD is the furthest along, others are also making progress in this area. COMPASS Pathways has achieved promising Phase 2 results with COMP360, the psilocybin therapy for treatment-resistant depression. Psilocybin is a naturally occurring psychedelic substance commonly found in ‘magic mushrooms’.
MindMed (MNMD) is involved in Project Lucy, currently in Phase 2, which uses doses of LSD to treat anxiety. LSD microdosing for ADHD is also being investigated.
For Atai Life Sciences (ATAI), psychedelic drugs make up five of the 10 candidates in the company’s pipeline, including MDMA, ketamine and psilocybin.

